How to get a Drug Distribution License in India?

Japsanjam Kaur Wadhera | Updated: Jan 22, 2021 | Category: Drug
Availability of drugs and medicines is necessary to ensure good health of the people and the government regulates the distribution of drugs and medicine in India to ensure than such goods are not abused or misused by any individual. The right to manufacture and sell drugs has to be done cautiously and the government has made strict rules for granting license for manufacturing and selling drugs. Drug distribution license in India is therefore required to be obtained in accordance to the provisions of the Drugs and Cosmetics Act, 1940. This article will provide you with information regarding how to get a drug distribution license in India.
Table of Contents
Defining “Drugs” under Drugs and Cosmetics Act, 1940
According to Section 3(b) of the Drugs and Cosmetics Act, 1940 “drug” means all the medicines and devices for the use of animals or human beings externally or internally and all the substances planned to be used in or for treatment, mitigation, diagnosis or to prevent any disease or disorder in human beings or animals including preparations applied on the human body for the purpose of repelling insects like mosquitoes and also includes all the substances that can be used for the purpose of destruction of insects and all components of drug like empty gelatine capsule. Later, in 1964, the definition was amended to include Unani drugs and Ayurvedic drugs.
The Central Drugs Standard Control Organization (CDSCO) and State Drug Standard Control Organization (SDSCO) are the regulatory body for the drug registration in India and has the power to issue drug distribution license in India. CDSCO is the head official of the Drugs Controller General India (DCGI).
It is necessary for every manufacturer of the cosmetic products to ensure and comply with the rules specified for registration and all other mandatory requirements under the Drugs and Cosmetics Rule 1945.
Conditions to obtain Drug Distribution License in India

Competent individual/Pharmacist
The pharmacist should be qualified in the case of retail business. In the case of wholesale business the person must be graduated with 1 year experience or undergraduate with 4 years experience.
Space/ Area of Pharmacy
Another condition to obtain drug distribution license in India is the requirement of space that is the area of the pharmacy/unit. For the purpose of wholesale and retail license, the unit must be 15 square meters. And in all other case, it must be 10 square meters.
The clear height of the premises where sales shall take place must be as per the guidelines laid down under the National Building Code of India, 2005.
Storage Facility
It is necessary for every pharmacy to have a storage facility. Since some drugs are required to be stored in low temperature, so the air conditioners and refrigerators are must.
Compliance
It is mandatory for the license holder is required to comply with the conditions of the license of the issuing authority.
Technical Staff hiring
It is mandatory to hire qualified personnel as staff at the medical ships or pharmacy.
How to obtain Drug Distribution License in India?
To obtain drug distribution license in India, the below mentioned steps are required to be followed: –
- The first step to apply for drug distribution license is that the applicant is required to apply online for the license on the basis of the line of business for which he must have a valid contact number and email id.
- Further, the applicant is required to keep all the documents updated and upload the form along with the documents and the prescribed fees.
- Once the online process is completed, the inspection shall take place by the inspector who shall visit the site and verify the validity of the documents.
- The authority shall issue the drug license to the applicant once all the above steps are duly complied.
Documents required in obtaining Drug Distribution License in India
The following documents are required to be submitted to obtain drug distribution license in India: –
- Memorandum of Association (MOA) and Articles of Association (AOA) for a company and a partnership deed, and in case of partnership and LLP an LLP agreement.
- ID proof of the proprietor, partner, director.
- If the premises is rented then rental agreement as the case may be.
- If the premises is self-owned then the copy of the ownership documents as the case may be.
- Key plan and site plan of the premises.
- Proof of the availability of the storage space as cold storage that is, refrigerator, air conditioners etc.
- Copy of the board resolution approving to obtain drug distribution license.
- Challan copy as a proof of depositing fee.
- An affidavit from the competent person or registered pharmacist.
- An affidavit regarding non-conviction of partner, proprietor or director of the firm.
- For a pharmacist in the wholesale sale: experience certificate, qualification proof and appointment letter.
- For a pharmacist in the retail sale: qualification proof, local pharmacy council registration and appointment letter.
What are the types of Drug licenses in India?
The different types of Drug Licenses under the Drugs and Cosmetics Act in India are as follows:
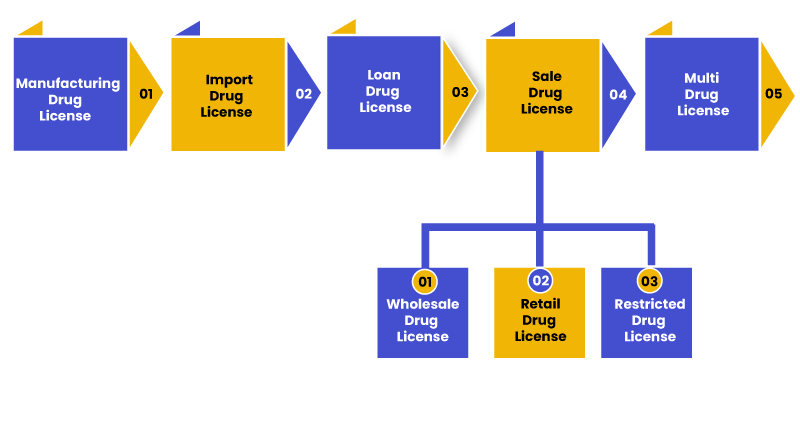
Manufacturing Drug License
All the manufacturers of the Ayurvedic, Cosmetic, Allopathic products or any other drug under the Drugs and Cosmetics Act 1940[1], is required to obtain manufacturing drug license. The government grants a drug license to the business in the state in which the manufacturing activity is performed.
Import Drug License
The businesses that distribute imported drugs in India or use imported products to manufacture drugs in India are required to obtain import drug license in India.
Loan Drug License
Those businesses which do not have their own land to manufacture its own drugs but want to manufacture the products with their brand name on the land of a business which already has a manufacturing drug license, are required to obtain or issue loan drug license in India.
Sale Drug License
All the retailers and wholesalers engaged in distributing drugs in India are required to issue or obtain sale drug license in India. It is further bifurcated in Wholesale drug license, Retail drug license and Restricted Drug License.
Wholesale Drug License
This license is issued by the Central Drugs Standard Control Organization to those wholesalers who are doing business in pharmaceuticals. In order to obtain this license, the person doing such business must hold a diploma or degree in Pharmacy from a reputed and recognized university and must have minimum of 1 year experience in dealing with drugs.
Retail Drug License
This license is issued by the Licensing Authority to retailers who are doing business in stand-alone pharmacists or pharmaceuticals. It is necessary for the applicant to be registered as Pharmacist with the State Pharmacy.
Restricted Drug License
This type of license is issued under Forms 20A and 21A to the persons or dealers to sell the drugs without the supervision of any qualified person.
Multi Drug License
Those businesses that have operations in more than one state are required to obtain Multi drug license. It is advisable to consult business lawyers in India to issue multi drug license in different states so as to avoid any penalties from the state authorities.
Conclusion
The manufacturing and selling of drugs are governed by the laws laid down by the Government of India. The process to get a drug distribution license in India can be very complicated as it is important to apply for the right kind of drug license and apply all the necessary documents along with it as required. The Drugs and Cosmetics Act, 1940 is a disciplinary act. It ensures that every person doing the business of drugs and cosmetics must follow all the rules and those not complying with it must be held liable to be punished for committing such offence.
Also, Read: Start Pharmaceuticals Export Business: Concept and Licenses Required














