Procedure for obtaining Drug License online in India

Khushboo Priya | Updated: Apr 29, 2019 | Category: Drug
I f you wish to sell medicines or open a pharmacy in India, firstly, you need to obtain a drug license. The reason why drug license is mandatory is to ensuring public health. Further, it is to ensure that medicines and related items are not abused by any entity within the Nation. Therefore, keeping all such things into consideration, the Government of India came up with strict laws for manufacturing and distributing medicines and drugs in India.
Drugs and Cosmetics Act, 1940 is the concerned law for regulating the manufacturing and sale of medicines or drugs and rules made thereunder. For the rest of the details regarding the drug license, we recommend you continue reading the blog. We will help you with every significant aspect of acquiring the license.
Table of Contents
Market Overview of Drug Industry or Pharmaceutical Industry
As per the latest report of IBEF, India is the world’s largest contributor to generic drugs. Moreover, the pharmaceutical industry of India supplies over 50% of global demands, for the number of vaccines. Besides, the Indian pharmaceutical sector provides over 80% of the antiretroviral drugs used for combating Acquired Immune Deficiency Syndrome (AIDS).
1. The international market scenario of the drug industry
Here’s the past, current, and future market scenario of Indian Drug Industry to the international market:
- In the year 2017, the value of the pharmaceutical industry was expected at US$ 33 billion.
- However, over 2015-20, the nation’s drug industry is estimated to increase at a CAGR (Compound Annual Growth Rate) of 22.4% to reach US$ 55 billion.
- Although pharmaceutical exports remained at US$ 15.52 billion in the Financial Year 2019 (up to January 2019).
2. The domestic market scenario of the Indian Pharmaceutical Industry
Not only in the international market but the Indian Drug Industry has achieved a hike in the domestic market as well. Here’s the actual market overview:
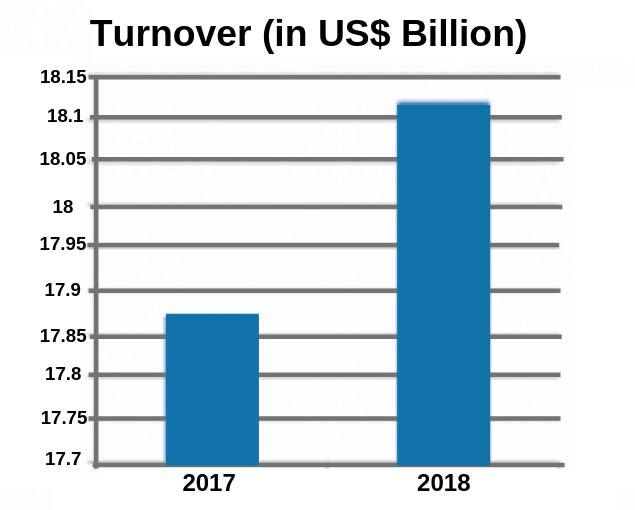
- In 2018, the turnover of India’s domestic pharmaceutical market reached to Rs. 129, 015 crore .i.e., US$ 18.12 billion. It has a growth of 9.4% as compared to the year 2017.
- In 2017, the pharmaceutical sector was calculated to be Rs. 116, 389 crores (US$ 17.87 billion).
What is Drug License?
The drug license is a mandatory permit granted by the governing authority under the Drugs and Cosmetic Act, 1940 to carry on the business of medicines or drugs or cosmetics. The concerned authority for issuing the license is the Central/State Drugs Standard Control Organization.
Types of Drug License in India
Mainly, there are three types of drug licenses issued in India. They are as follows:
- Manufacturing License
- Loan License
- Sales License
In this write-up, we will primarily focus on the sales license. The sales license is further classified as:
- Retail license
- Wholesale license
- Restricted license for general stores (which depends on the type of sales done)
Checklist for obtaining the drug license in India
In order to obtain a drug license in India, you need to satisfy the following conditions:
-
For competent person/pharmacist
There must be a specialist in medicines who can take care of all the drugs. Additionally, if you are applying for a retail license, then you should be a qualified pharmacist. However, if you need a wholesaler license, you should be a graduate with at least one year of experience or an undergraduate with four years of experience.
-
Area of the pharmacy set-up
The required area of the pharmacy shop should be at least 10 square meters. Besides, if you are applying for both wholesale and retail pharmacy, the minimum area of the carpet must be 15 square meters. Furthermore, the premises must contain ventilation.
-
The storage facility of drugs
One must store the medicines in a clean, hygienic, cool, and ventilated area. However, certain drugs and vaccines require a low-temperate freezing zone for storage. While an air conditioner and a refrigerator are mandatory for everybody willing to acquire a drug license in India. Therefore, every medicine and drug should be stored as prescribed or the authority will reject the application for the license.
Documents required for obtaining a license for selling drugs in India
You require providing the following documents for acquiring sales license for selling the drug in India:
- Memorandum of Association (MOA) and Articles of Association (AOA) of the firm;
- Identity proof such as PAN card of the sole proprietor, directors and their partners;
- Address proof of the owned office premises;
- In the case of rented property, rent agreement is mandatory;
- Details of drug storage such as invoices of the refrigerator and air conditioner;
- Qualification proof such as certificates or form of degrees or mark sheets;
- Any other additional wholesale documents for obtaining a wholesale license for selling drugs;
- Required experience certificate depending upon the kind of sales license;
- Other licenses as required.
Procedure for obtaining drug license online in India
For acquiring a drug license, you need to file an application with the concerned authority of your state. However, before that, you need to find out the form suitable for you. Below, we have described a few significant steps to help you with online registration. Here we go:
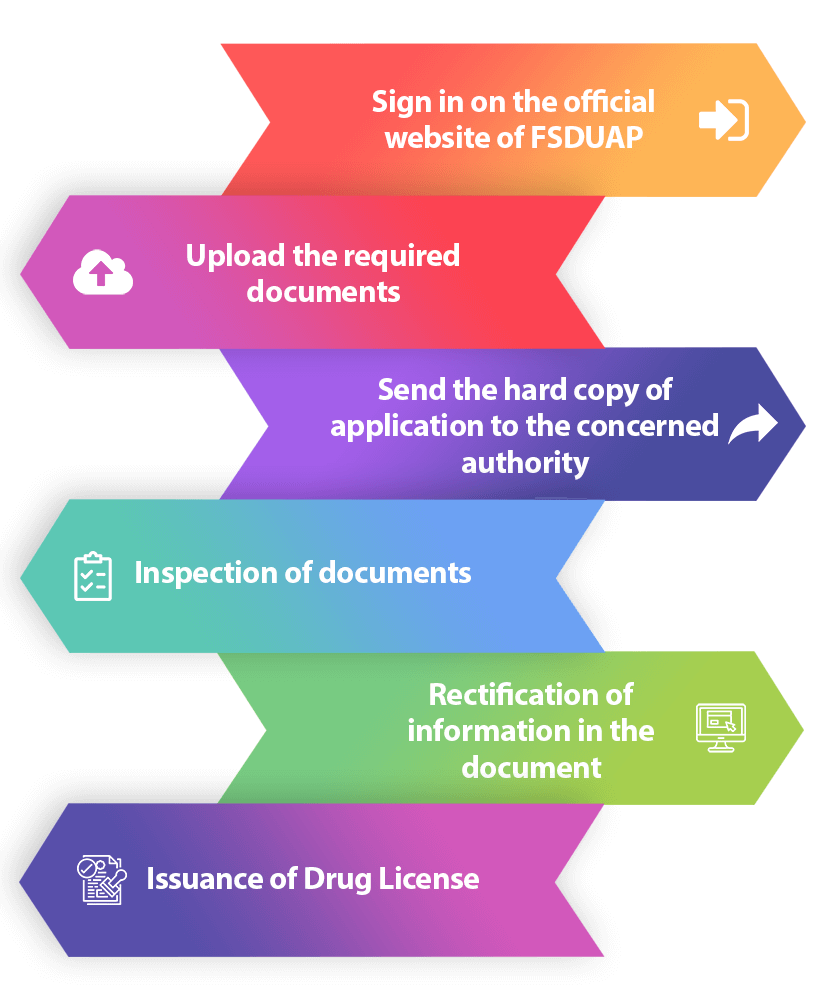
Step 1: Sign in on the official website of FSDAUP
Firstly, go to the official website of FSDAUP, and sign in as a new user. Fsduap is an online platform for the Food Safety and Drug Administration. There, you need to select the option of online registration for Drugs/Cosmetics sales.
Step 2: Upload the required documents
Secondly, upload the essential documents required for drug license for sales and click on submit.
Step 3: Send the hard copy of application and documents to the concerned authority
Next, you require sending one hard copy of the application along with all the other obligatory documents to the office of Food Safety and Drug Administration.
Step 4: Inspection of documents
Once you have sent the documents to the concerned authority, they will scrutinize them as intimated to the applicant.
Step 5: Rectification of information in the document
In the case, the Drugs Control Authorities find any error; they will give the applicant a prescribed time for rectification.
Step 6: Issuance of a Drug license
Once the applicant has addressed the objection, he/she will receive the license from the Licensing Authority.
Hence, this is all the registration process of the drug license required. However, if you still have some doubts, you can leave a comment below. Or if you want a drug license, you can contact Swarit Advisors. We will aid you with the fastest issuance of the license.














