Overview of Drug Export NOC
In India, the pharmaceutical industry is on the top of the list worldwide. India ranks thirteen in terms of value and ranks third in volume. India plays a significant role in supplying generic medicine to many countries worldwide. India has emerged as a drug-manufacturing hub and research self-starter for many years. Here has been a continuous
growth in the export of pharmaceutical products from India, and it is becoming a new professional opportunity for many. If we see the tendency, generic drug exports dominate nearly 70% of the world market, whereas patented drugs and over the counter drugs make up 21% and 9%, severally. The top countries importing laboriously from India are the USA, UK, South Africa, Russia and Nigeria. India is the world's biggest exporter of generic drugs. The manufacturing price of generic drugs is much lower in India than in the USA and Europe.
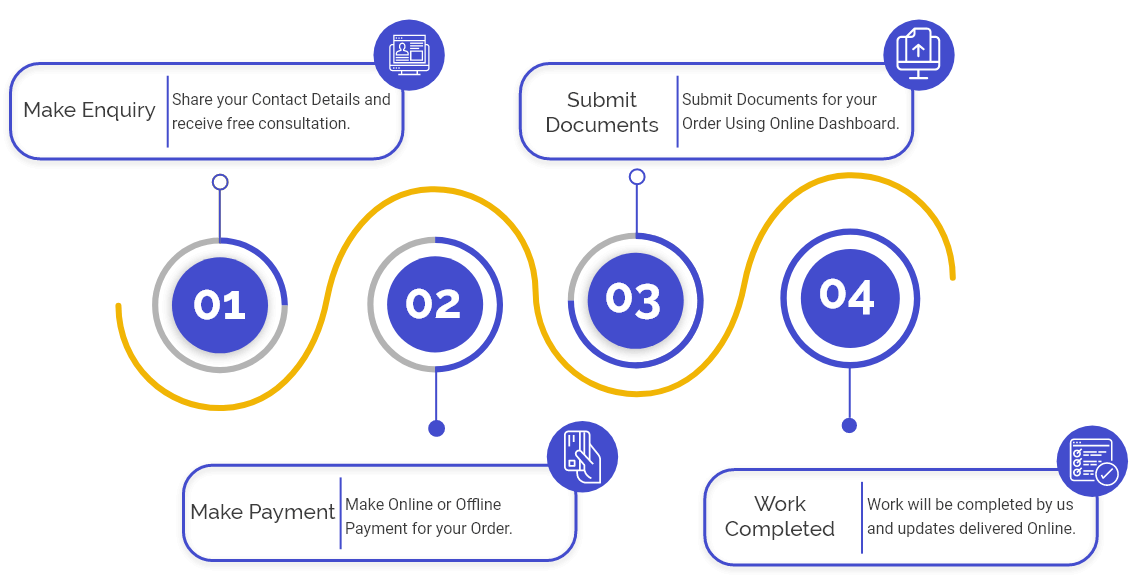
Exporting medical-related products requires a NOC (No Objection Certificate) from the Drug Controller Office of India. NOC is a document along with the Drug Controller that permitted manufacturers of medical products to export their Pharmaceutical products freely outside India. In India, the manufacturer holds a valid License to manufacture drugs according to Form 25 and Form 28 and can obtain a Non-objection Certificate. CDSCO (Central Drugs Standard Control Organisation) has authorised State Licensing Authority (SLA) to issue a NOC to export only Approved/Unapproved and banned drugs in India from 20 August 2018. An entity seeking NOC can apply to regional or sub-regional offices of the CDSCO.
Essence for applying Drug Export NOC
For getting NOC for approved or unapproved and banned drugs, the manufacturer must have a valid license to manufacture pharmaceutical products in India. Apart from this vital requirement, we must consider the following conditions.
- The drug export application should provide a valid export order, and a NOC will be granted for each order on a case-to-case basis.
- Applicants need to secure that the product for which NOC is liable is only for export, and by no possibility it will be rerouted for sale in the domestic market.
- The applicant must identify the site where the drug for export will be manufactured. They also need to submit whether the batch to be exported undergone quality control testing.
- Consignment information dispatched, stock remaining, raw material, and intermediates must be made available for inspection by the appropriate department.
- The applicant for NOC should prove that there will be physical destruction of all residue drugs meant for export only.
- The candidate is also required to ensure that will stop the production or export of such drugs; in the future, drugs fall into the classification of prohibited medications in the home country or importing country.
- The candidate must also submit all essential documents in a standard submission Format for obtaining NOC.
Documents Required for Drug Export NOC
Following are the documents required for obtaining Drug Export NOC.
- Covering Letter: it is a vital part of every application, and it identifies
- the intent of the applicant. The covering letter has a list of documents and other details necessary to be submitted. The letter must recognise the name of the drugs, composition, dosage and power of the medications, along with the country's name and cost to be exported. The authorised witness must duly sign the documents required. Must also mention the authorised witness's name and designation.
- Purchases Order: valid copy of the purchase order from the overseas buyer in the name of the manufacturers or trader in which a list of products to be exported is mentioned needs to be submitted. The order must carry the name of the drug, composition, dosage and the quantity of the product required. The competent witness authority must duly sign the acquire order, and it must have an at the latest date, not more than six months before the application made by the firm.
- Manufacturing License: the copy of the license granted by the competent authority to manufacture drugs for export must be enclosed with the application.
- Performa Invoice: a correctly signed copy of the Performa invoice from the importing country must surround the application. An invoice should be attached with the application to import unapproved active pharmaceutical components.
- Registration Certificate: (Applicable for banned drug)Registration Certificate from the National Regulatory Authority of importing country.
Ministry of Health and Family Welfare has provided particular rules and guidelines for the export of drugs, which manufacturer has to comply with;
- Aadhar Card
- Residence Proof;
- Age Proof;
- Applicant Bank account details such as account number, bank branch, and IFSC/RTGS Number;
- Details of authority & employer’s certificate of working for 90 days as construction labour & nomination form.
How will SWARIT Advisors help you?
SWARIT Advisors has dedicated terms of compliance and regulatory professionals who can help provide end-to-end service for obtaining import Licenses. Our expert and experienced professionals ensure our client's excellent services of lengthy compliance processes. We gave on-time delivery of government registration and licensing. Facilitate comprehensive assistance throughout the Registration process, and hassle-free gets Drug Export NOC.
Frequently Asked Questions
The CDSCO regulates the export and import of drugs in India. There are 11 ports in different parts of the country where export and import are done. CDSCO regulates India's sales, manufacturing, and clinical research, apart from export and import.
NOC is a legal document granted by any individual, organisation, or agency to not object to the matter mentioned in the certificate.
A unique code or natural code needs approval by CDSCO. It is necessary when the manufacturer does want its name and address to reflect on the label. To get approval for a unique code for the finished formulation or bulk manufacturer need to apply to get the code.
Documents necessary for applying a unique or neutral code:
- Correctly signed application for drug export NOC on the Company's letterhead.
- Copy of valid Export Order identifies the name and the amount of the drug to be exported.
- Letter from international buyer specifying the requirement of a particular code number.
- Copy of NOC for Export granted by State Licensing Authority for specific product packing material, including labels, leaflets, foils, cartons etc., specifying the unique code.
- A valid copy of the Registration certificate granted by the Ministry of Health of the importing country is provided.
According to our Foreign Trade Policy, only registered pharmaceutical companies can export medicines from India. Primarily, the Company must register with the Director-General of Foreign Trade and apply for an Import Export code.
A duly attested and valid copy of a wholesale License for the sale/ issuance of drugs under Drugs and Cosmetics Rules in form-20B & 21B or its renewal in form-21C granted to the manufacturer or its representative by the State Licensing Authority in India.